 The whole isn’t always the sum of the parts concerning the relationship between copolymers and their monomers. Some monomers have an outsized influence on polymer properties. One monomer that can bring a unique set of properties when used as in a copolymer binder for coatings is Isobornyl Methacrylate or IBOMA. The use of IBOMA in coatings is not limited to polymers, it also can be used in radiation cured coatings as a reactive diluent and in oligomers for 2K and UV cured coatings. Let’s explore some of the unique properties IBOMA brings to coatings.
The whole isn’t always the sum of the parts concerning the relationship between copolymers and their monomers. Some monomers have an outsized influence on polymer properties. One monomer that can bring a unique set of properties when used as in a copolymer binder for coatings is Isobornyl Methacrylate or IBOMA. The use of IBOMA in coatings is not limited to polymers, it also can be used in radiation cured coatings as a reactive diluent and in oligomers for 2K and UV cured coatings. Let’s explore some of the unique properties IBOMA brings to coatings.
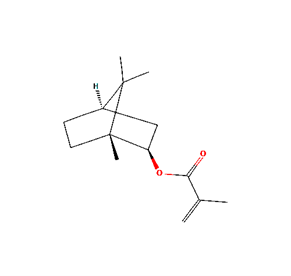
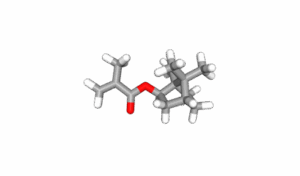
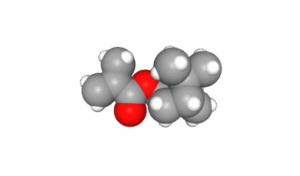
Isobornyl Methacrylate (CAS number 7534-94-3) is a saturated, bicyclic organic compound with the empirical formula C14H22O2 and a molecular mass of 222.32 grams per mole. The IUPAC name is [(1R,2R,4R)-1,7,7-trimethyl-2 bicyclo[2.2.1]heptanyl] 2-methylprop-2-enoate but is commonly known as Methacrylic acid Isobornyl ester and IBMA, we will use the previously mentioned common name, IBOMA. IBOMA has low volatility which makes it ideal as a monomeric low VOC diluent in radiation cured systems. The large bicyclic side chain contributes to the unique properties of copolymers containing IBOMA. Various depictions of the structure of IBOMA are shown below. The IBOMA structures are courtesy of the NIH PubChem website1.
ULTRUS Collection solve customer problems across product stewardship, ESG, renewable energy, learning and workplace safety. Learn more here!
A homopolymer of IBOMA has a very high glass transition temperature (Tg), so polymer chemists consider it a “hard” monomer, meaning it imparts hardness to copolymers containing it. The homopolymer of IBOMA has a very high glass transition temperature at 110.5° C which is harder than homopolymers of methyl methacrylate or Styrene. These monomers are traditionally used when a hard monomer is needed to make a high Tg copolymer. The Tg of a copolymer is calculated using the Fox Equation shown below. This demonstrates that even small amounts of a high glass transition temperature monomer can have a large effect on the Tg and other polymer properties.
Tg =Tg 1+Tg 2(+…)
Where:
- = glass transition temperature of the copolymer (in Kelvin)
- = glass transition temperatures of the homopolymers (Kelvin)
- = weight fractions of each monomer (must sum to 1)
IBOMA copolymerizes well with commonly used monomers like Butyl Acrylate, 2-Ethylhexyl Acrylate, Methyl Methacrylate, Styrene, Hydroxyethyl Acrylate, Acrylic Acid, and Glycidyl Methacrylate. The reaction rate of IBOMA is well balanced, the methacrylate group reacts quickly but the reaction is moderated by the steric hindrance of the bulky Isobornyl group. Coatings properties IBOMA copolymers bring to the table include hardness, high gloss, clarity, and UV resistance. Unlike hard polymers made with methyl methacrylate or styrene, those using IBOMA are not brittle. Let’s look at what features of IBOMA’s structure contribute to these properties.
The flexibility of IBOMA copolymers enhances coating flexibility making it more resistant to cracking and crazing. The increased flexibility of IBOMA containing polymers enhances the adhesion of coatings formulated from them. Stronger interactions between coating and substrate are made possible improved substrate wetting. The bulky Isobornyl group increases intermolecular attraction between the coating and surface by amplifying the influence of Van der Waals forces. This adhesion improvement occurs with various materials including metal, wood and plastic. Improved adhesion may eliminate the need for primers and increase the performance of a coating in other areas such as corrosion and moisture resistance.
Solvent based coatings polymers containing IBOMA typically have lower viscosity than those made from other monomers at comparable solids content and molecular weights. The steric hindrance of the non-polar, bulky side chain group reduces intermolecular interactions such as hydrogen bonding. The monofunctionality of IBOMA does not inherently increase cross link density. This enables lower VOC paint formulations with higher nonvolatile content at the same application viscosity. The advantages of higher solids coatings include lower pollution from evaporating solvents, a cleaner and safer working environment for workers, and higher dry film thickness per coat during application.
The large, saturated bicyclic side chain on IBOMA does not absorb UV energy like the aromatic side chain found, for example, on Styrene. IBOMA, when polymerized, also lacks conjugated double bonds which can also absorb UV energy. The absorption of UV energy leads to excited states and possibly bond scission. If bond breaking leads to free radical formation, a self-perpetuating chain reaction can occur causing degradation of the coating film. Since IBOMA is highly hydrophobic, there is less water absorption minimizing defects such as environmental stress cracking. The aliphatic structure of IBOMA doesn’t yellow like aromatic high Tg monomers.
Clearly, when used in polymers for coatings IBOMA’s structure is more than the sum of its parts. IBOMA has many other advantages. The monomer can be readily made commercially from non-food chain, renewable raw materials such as camphor or pinene. VISIOMER® TERRA IBOMA2, manufactured by Evonik, has a Certified Bio-Content of 72% and contributes to the overall biobased content of copolymers containing it. The unique structure of IBOMA enables high performance and sustainability attributes to coatings made from IBOMA copolymers.
1https://pubchem.ncbi.nlm.nih.gov/compound/Isobornyl-Methacrylate
2 https://www.evonik.com/en/products/oa/pr_52037624.html
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.
