
Titanium dioxide (TiO2) is the most used white pigment for paints and for other systems. It gives a coating whiteness and opacity (= hiding power)1,2. Especially when volume prices of systems are used, the pigment is considered as being expensive. The high volume price of TiO2 is related to its high density.
Because of its high price, it is of interest to use TiO2 as efficient as possible.
Scattering by solid particles
Particles in a matrix, like a binder system in a coating, change the direction of light when the particles and the medium have a different refractive index (n). This phenomenon, called scattering, results in both white color and hiding power of the coating3.
A pre-condition for obtaining bright whiteness is that the particles do not absorb visible light, electromagnetic radiation with wavelengths (l) between, roughly, 380 and 700 nm. Pure titanium dioxide complies with this demand.
A few key properties govern the scattering efficiency of particles in a medium.
ULTRUS Collection solve customer problems across product stewardship, ESG, renewable energy, learning and workplace safety. Learn more here!
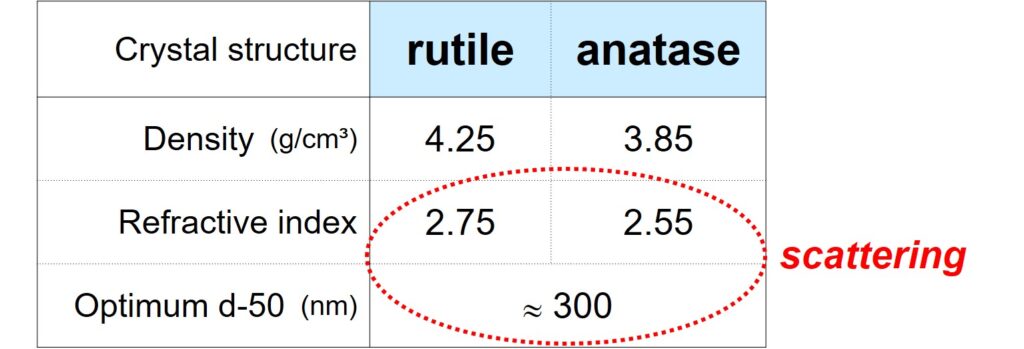
First, scattering is strong when the difference in refractive index of particle and matrix, Dn = np – nm, is big. For binders used in coatings, the refractive index is around 1.55. TiO2 is preferably used as scattering source because the pigment has a high refractive index, that depends on the crystal structure of the pigment.
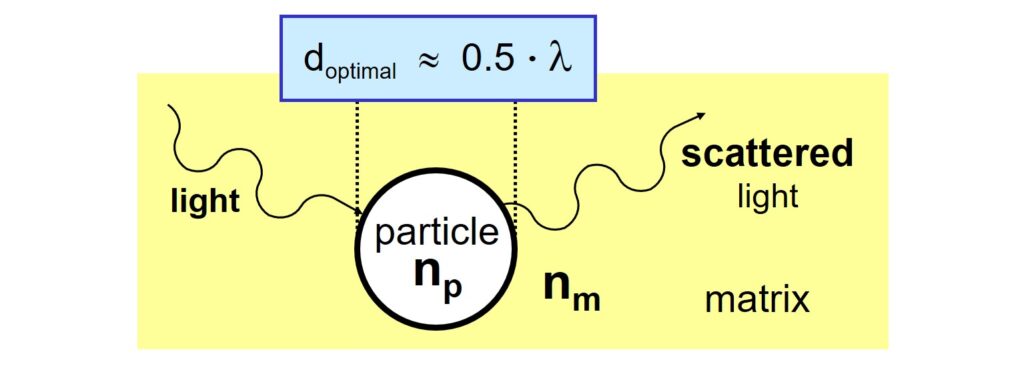
Secondly, for a specific wavelength of light, l, there is an optimum particle size. The diameter (d) of the particles should be close to half of the wavelength of the light for maximum scattering.
This implies that rutile TiO2 particles with a diameter of around 300 nm scatter visible light most efficiently.
Optimizing the scattering efficiency of TiO2
After having selected the right TiO2 pigment, with respect to purity, crystal structure and particle size, several approaches can be used to maximize the efficiency of expensive TiO2 pigment.
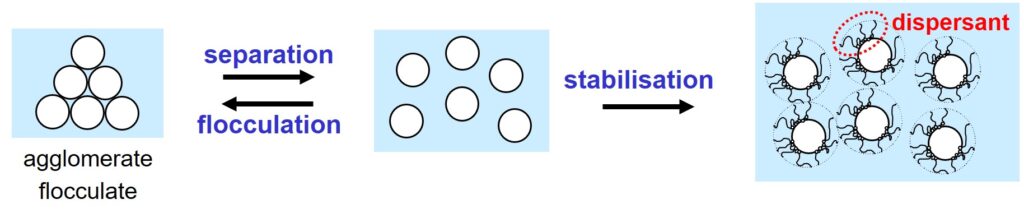
First, during the dispersion process, the primary pigment particles must be separated from each other as much as possible. Also, the particles must be stabilized with dispersant to prevent flocculation4,5.
A considerable amount of money can often be saved by improving the separation process and/or by changing the type and/or amount of dispersant.
Secondly, it is important that the TiO2 particles are distributed uniformly over the complete system. This phenomenon is called spacing. An approach to prevent crowding, the opposite of spacing, is to use a suitable filler. The objective is that filler particles fill the spaces between the TiO2 particles, thus acting as spacers.
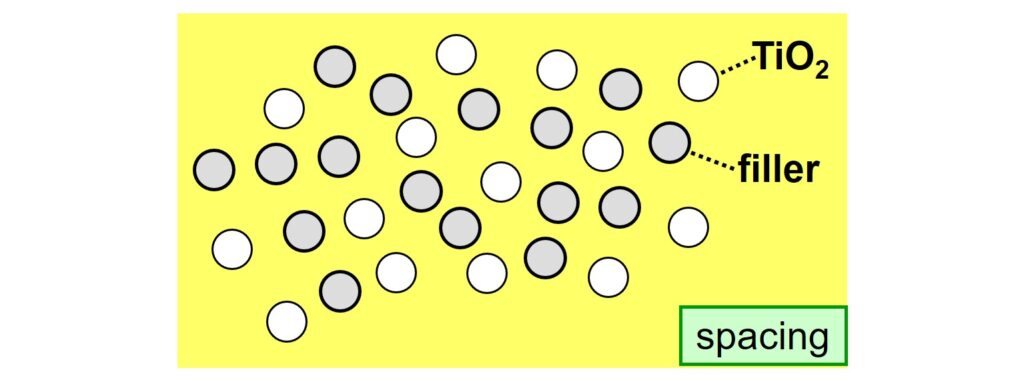
For a filler to provide spacing, the particles must be small enough and, preferably, the filler has a low color to minimize absorption as much as possible. An example of a spacer filler that complies with both criteria is OptiGlossTM XD from Imerys. The average particle diameter (d-50) of this bright kaolin filler is around 200 nm.
Another concept that can be used to obtain maximum scattering power is to use TiO2 particles that have a thick layer of post-treatment. Ti-Pure™ TS-6300 Chemours is a white pigment that consists of rutile TiO2 cores that are post-treated with a thick layer of oxides. The diameter of the TiO2 cores is in the range of 300 nm, and the average diameter of a complete particle is 530 nm. The thick oxide shell provides spacing: the TiO2 cores cannot come close to each other because of the post-treatment.
References
- Titanium Dioxide: An Introduction, Marc Hirsch, 23 September 2016.
- Titanium Dioxide: The Misunderstood Pigment, Marc Hirsch, 27 March 2020.
- Optical Properties of Pigments: Absorption and Scattering, Jochum Beetsma, 9 March 2022
- Understanding Dispersants, Marc Hirsch, 19 February 2016.
- The Basics of Dispersion and Stabilization of Pigments and Fillers, Jochum Beetsma, 12 February 2025
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.
