 by Ronald Lewarchik & Vishakha Makode
by Ronald Lewarchik & Vishakha Makode
Exterior architectural coatings do much more than provide color. They protect building materials from weather, moisture, sunlight, and environmental contaminants while maintaining the visual appearance of the structure. As building designs continue to evolve and sustainability becomes a larger part of construction planning, coatings must provide both durability and moisture management.
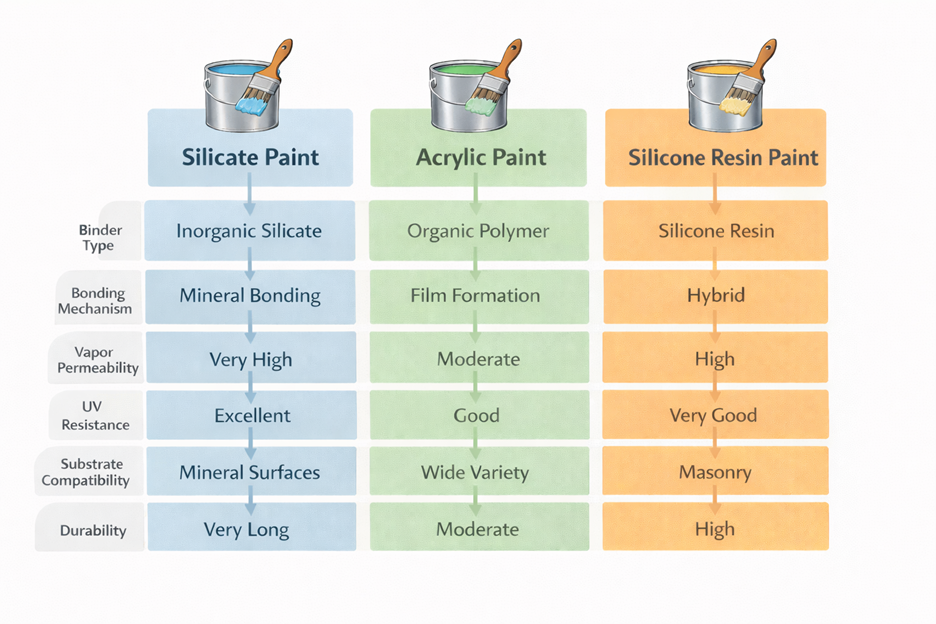
Most architectural coatings used today are based on organic polymers such as acrylics, vinyl, or silicone resins. These materials form protective films that cover the substrate and shield it from environmental exposure. While polymer coatings perform well in many applications, they can sometimes limit the movement of moisture vapor through masonry systems. Over long periods of outdoor exposure, ultraviolet radiation and environmental pollutants can also affect polymer coatings.
Mineral coatings offer a different approach. Instead of forming a flexible polymer film, silicate paints rely on chemical interaction with the substrate itself. This technology has been used in architectural coatings for well over a century and remains widely used for masonry and concrete surfaces.
When applied to a mineral surface, the silicate binder penetrates the porous structure of the substrate and reacts with the minerals present in masonry materials. At the same time, the binder slowly reacts with carbon dioxide from the surrounding air. These reactions gradually produce a mineral silica network that becomes integrated with the substrate.
In an earlier article published in the UL Prospector Knowledge Center, “Waterborne Silicate Coatings: The Ultimate Eco-friendly Coating,” the chemistry and environmental advantages of silicate coatings were discussed in detail. That article explained how these coatings are derived from silica and alkali metal compounds such as lithium, sodium, or potassium silicates. The article also described the environmental advantages of silicate coatings, including their durability, ultraviolet stability, chemical resistance, and low VOC content.
The present discussion expands on those concepts and focuses on the formulation and practical use of silicate façade coatings in modern architectural applications.
Silicate Binder Chemistry
Silicate paints are based on alkali silicate binders commonly referred to as waterglass. These materials consist of aqueous solutions containing silicate structures combined with alkali metal ions such as potassium or sodium.
Among these options, potassium silicate is generally preferred for exterior architectural coatings because it provides good weathering resistance and interacts well with mineral substrates.
When a silicate coating is applied to concrete or masonry, the liquid binder penetrates the pores of the surface. This allows the binder, along with pigments and fillers, to move into the upper layers of the substrate.
As the coating begins to dry, the silicate binder undergoes polymerization reactions. At the same time, it reacts with minerals present in the substrate. In cement-based materials, calcium hydroxide present in the matrix can react with the silicate binder.
Another important reaction occurs with carbon dioxide in the air. This carbonation process gradually converts soluble silicate into insoluble silica. As these reactions progress, a mineral network forms that anchors the coating to the surface.
Because the coating becomes chemically integrated with the substrate, silicate paints often show exceptional durability and resistance to ultraviolet degradation.
ULTRUS™ Product Stewardship enables material selection, compliance, sustainability and chemical management throughout the product life cycle to meet regulatory requirements and consumer demands. Learn more here!
Silicification Mechanism
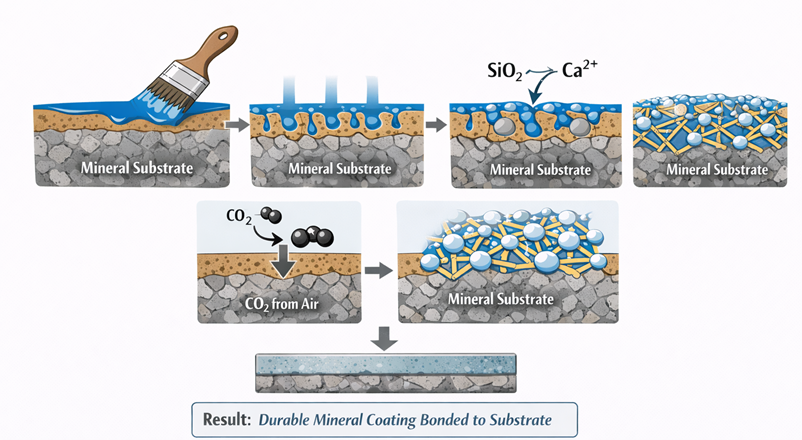
The curing of silicate paints occurs through a sequence of chemical and physical changes commonly described as silicification. This process is responsible for the strong bonding that develops between the coating and the mineral surface.
The process generally occurs through several stages.
First, the liquid silicate coating is applied to the mineral surface and as it is waterborne and low in viscosity, it penetrates into the pore structure of masonry or concrete.
Once inside the pores, the silicate binder begins reacting with calcium-containing minerals in the substrate. At the same time, the alkaline binder gradually reacts with carbon dioxide from the atmosphere.
As these reactions proceed, an insoluble silica network forms. This network binds the silicate film former along with the pigment and filler to the mineral surface and produces a coating that becomes an integral part of the substrate surface rather than remaining a separate film.
This mineral bonding mechanism is one of the main reasons silicate coatings are known for long service life and strong weather resistance.
Modern Variations of Silicate Coatings
Over time, several variations of silicate coating technology have been developed. These systems provide greater flexibility while maintaining the fundamental characteristics of mineral coatings.
Pure Silicate Coatings
Pure silicate paints rely almost entirely on mineral binders and inorganic pigments. These coatings provide excellent vapor permeability and long-term durability. However, they require compatible mineral substrates to perform properly. Pure silicate coatings are commonly used on new masonry construction and in historic building restoration projects where the mineral surface of the substrate is cleaned of any efflorescence or other surface issues.
Dispersion Silicate Coatings
Dispersion silicate paints contain a small amount of organic polymer dispersion. This modification improves adhesion when coatings are applied to renovation surfaces or previously painted substrates. Although a small polymer fraction is present, these coatings still maintain the breathable properties associated with mineral coatings.
Sol-Silicate Coatings
Sol-silicate coatings combine potassium silicate with colloidal silica. This combination creates a hybrid binder system that can improve adhesion and penetration on dense mineral surfaces. Sol-silicate coatings are widely used today because they provide greater formulation flexibility while preserving the advantages of silicate technology.
Pigments and Fillers in Silicate Formulations
The alkaline nature of silicate film formers place certain limitations on pigment selection. Many organic pigments are not stable in high pH environments and may degrade over time. For this reason, silicate paints typically use inorganic pigments such as iron oxides or mixed metal oxide pigments. These pigments provide good color stability and excellent resistance to ultraviolet radiation.
Mineral fillers are also important components in silicate formulations. Materials such as calcite, dolomite, silica flour, and mica help control opacity, rheology, and coating structure. Silicate paints often operate at relatively high pigment volume concentrations. This contributes to the characteristic matte appearance of mineral coatings while maintaining high vapor permeability. Because pigment and filler loading strongly influence coating performance, balancing pigment volume concentration with binder content is an important part of formulation design.
Example Silicate Façade Paint Formulation
The following table illustrates a typical starting point for laboratory development of a silicate façade coating.
| Component | Function | Typical Range (%) |
| Potassium silicate solution | Primary binder | 20–30 |
| Colloidal silica | Secondary binder | 5–10 |
| Titanium dioxide | Opacity | 5–8 |
| Iron oxide pigment | Color | 2–5 |
| Calcite / dolomite | Mineral extender | 20–35 |
| Silica flour | Structure modifier | 5–10 |
| Mica | Lamellar filler | 3–6 |
| Additives | Dispersant / rheology | 0.5–1.5 |
| Water | Balance | remainder |
Balancing pigment volume concentration with binder content is essential. Excess filler can weaken the coating structure, while too much binder may reduce vapor permeability.
Substrate Preparation
Silicate coatings perform best on rigid mineral substrates such as concrete, brick masonry, cement render, and natural stone. These surfaces provide the mineral chemistry required for silicification.
Proper surface preparation is critical. The substrate must be clean, structurally sound, and free of incompatible coatings.
Highly porous surfaces may absorb water too quickly during application. To prevent this, a mineral primer or silicate fixative is often applied before the topcoat.
Applying a silicate primer or fixative before the topcoat helps equalize substrate absorption and improves coating uniformity.
Practical Performance Testing – Façade Coating
Developing a reliable silicate façade coating requires evaluating both coating performance and substrate interaction.
| Property | Test Method |
| Adhesion | ASTM D4541 |
| Vapor permeability | ISO 7783 |
| Water absorption | EN 1062-3 |
| Weathering durability | ASTM G154 |
| Abrasion resistance | ASTM D4060 |
| Scrub resistance | ASTM D2486 |
Façade coatings must balance vapor permeability with resistance to liquid water penetration. Both properties should be evaluated during formulation development.
Comparison of Façade Coating Technologies
Formulator Takeaways
- Substrate compatibility determines performance: Silicate coatings rely on chemical interaction with mineral substrates, making proper surface preparation essential.
- Pigment stability is critical: Only pigments capable of withstanding high alkalinity should be used in silicate paint formulations.
- Breathability must be preserved: Balancing binder content and pigment volume concentration ensures both durability and vapor permeability.
Conclusion
Silicate paints represent a distinctive category within architectural coatings. Their ability to chemically bond with mineral substrates provides durability and breathability that are difficult to achieve with conventional polymer coatings.
Advances in sol-silicate and dispersion silicate technologies have expanded the practical applications of mineral coatings while preserving their fundamental advantages.
As architectural design continues to emphasize durability, sustainability, and moisture management, silicate coatings will likely remain an important solution for modern façade protection.
References
- Waterborne Silicate Coatings: The Ultimate Eco-friendly Coating. UL Prospector Knowledge Center.
/9072/pc-waterborne-silicate-coatings-ultimate-eco-friendly-coating/ - Bonding and Coating Applications of PQ® Soluble Silicates – Technical Bulletin.
https://docslib.org/doc/8241480/bonding-and-coating-applications-of-pq%C2%AE-soluble-silicates - Waterborne Silicate Coatings. Rev. 3, July 2016.
https://books.google.com/books/about/Waterborne_Silicate_Coatings.html - Efflorescence of Soluble Silicate Coatings.
https://www.sciencedirect.com/science/article/pii/002230939190146W - American Chemical Society.
https://pubs.acs.org/doi/10.1021/bk-1982-0194
The views, opinions and technical analyses presented here are those of the author or advertiser, and are not necessarily those of ULProspector.com or UL Solutions. The appearance of this content in the UL Prospector Knowledge Center does not constitute an endorsement by UL Solutions or its affiliates.
All content is subject to copyright and may not be reproduced without prior authorization from UL Solutions or the content author.
The content has been made available for informational and educational purposes only. While the editors of this site may verify the accuracy of its content from time to time, we assume no responsibility for errors made by the author, editorial staff or any other contributor.
UL Solutions does not make any representations or warranties with respect to the accuracy, applicability, fitness or completeness of the content. UL Solutions does not warrant the performance, effectiveness or applicability of sites listed or linked to in any content.
